What Are the Degree of Polymerization and Degree of Hydrolysis of PVOH?
The properties of polyvinyl alcohol (PVOH, PVA, poval) are largely determined by two key parameters established during the manufacturing process: the degree of polymerization (DP) and the degree of hydrolysis (DH). The DP strongly influences properties such as aqueous solution viscosity and film strength, while the DH has a significant impact on water solubility, water resistance, and gas barrier performance. Understanding these two parameters is essential for selecting the PVOH grade best suited to a given application.
This article first outlines how PVOH is manufactured, and then explains the definitions, calculation methods, and effects of the DP and the DH on material properties, in an easy-to-understand manner. It is intended to help users identify which parameters to focus on when considering the introduction or selection of PVOH.

- Polymerization and Hydrolysis (Saponification) of PVOH: Explained from the Manufacturing Process
- Degree of Polymerization of PVOH ―A Key Parameter Affecting Viscosity, Strength, and Processability
- Degree of Hydrolysis of PVOH ―A Parameter Governing Water Resistance, Gas Barrier Performance, and Emulsification
- High Water-Soluble and Modified PVOH
1. Polymerization and Hydrolysis of PVOH: Explained from the Manufacturing Process
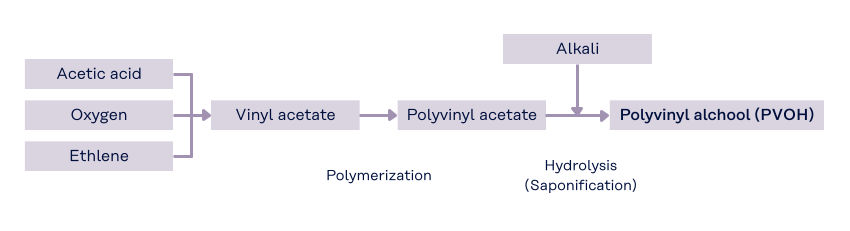
PVOH is produced through a two-step chemical process starting from vinyl acetate monomer (VAM), which is synthesized from ethylene, acetic acid, and oxygen.
- Polymerization → Formation of polyvinyl acetate (PVAc)
- Hydrolysis → Conversion of PVAc into PVOH
Vinyl Acetate Monomer (VAM) → Polymerization → Polyvinyl Acetate (PVAc) → Hydrolysis (Saponification) → Polyvinyl Alcohol (PVOH)
1-1. What Is Polymerization? — How the Degree of Polymerization Is Defined
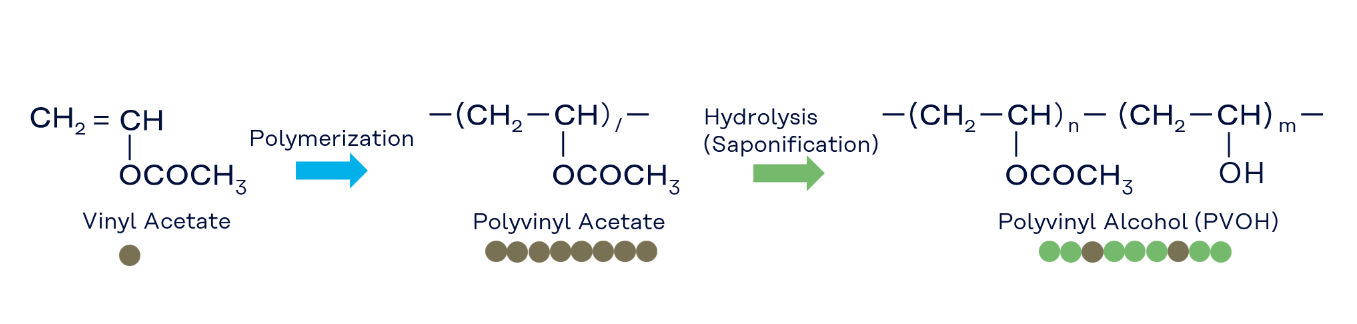
Polymerization is the reaction in which numerous vinyl acetate monomer molecules are linked together to form polyvinyl acetate (PVAc). Industrially, PVAc is typically produced by radical polymerization, using methanol as the main solvent.
The degree of polymerization (DP) represents the number of vinyl acetate units connected in the polymer chain. This parameter has a major influence on the properties of the resulting PVOH, particularly on aqueous solution viscosity and film strength. In general, a higher DP leads to higher viscosity and stronger films.
At the polymerization stage, it is also possible to copolymerize VAM with other monomers. Through such modification techniques, modified PVOH grades with properties different from those of conventional PVOH can be produced.
1-2. What Is Hydrolysis (Saponification)? — How the Degree of Hydrolysis Is Defined
Hydrolysis (saponification) is the reaction in which the acetyl groups (CH₃CO–) of PVAc are cleaved to form hydroxyl groups (–OH). PVOH is obtained through this reaction. Industrially, hydrolysis is typically carried out by alkaline hydrolysis, with methanol commonly used as the solvent.
By controlling how many acetyl groups are converted into hydroxyl groups during this reaction, PVOH grades with various degrees of hydrolysis (DH) can be produced. The DH has a profound effect on key properties such as water solubility, water resistance, and gas barrier performance (further details are explained in the next section).
Summary
PVOH is manufactured through a two-step process—polymerization followed by hydrolysis. The DP and the DH determined at these respective stages together constitute the core of PVOH grade design and enable optimization of properties for specific applications.
2. Degree of Polymerization of PVOH ―A Key Parameter Affecting Viscosity, Strength, and Processability
The degree of polymerization (DP) indicates the length of the molecular chain, i.e., how many vinyl acetate units are linked within a PVOH molecule. It can be calculated using the numbers of repeating units “m” (hydroxyl units) and “n” (residual acetyl units) as shown on the right.
The DP is controlled during the polymerization step and has a major impact on final product performance.
-1)
Degree of polymerization (DP)
n + m ... Indicates molecular chain length
Effects of the Degree of Polymerization on Properties
Differences in the DP significantly influence the following properties. Please refer to this information when selecting the appropriate grade for your application.
- Aqueous Solution Viscosity: As the DP increases, molecular entanglement becomes more pronounced, resulting in higher solution viscosity. Low-DP grades exhibit low viscosity, are easier to handle, can be dissolved at higher concentrations, and provide greater formulation and coating-process flexibility.
- Film and Mechanical Strength: The tensile strength and tear strength of PVOH films generally increase with higher DP.
- Film Formation Stability: High-DP grades readily form strong films but may require careful control of dissolution and viscosity adjustment. Low-DP grades dissolve easily and enable uniform film formation, making them suitable for low-viscosity formulations and thin-film applications.
Evaluation points:
aqueous solution viscosity, coatability, film and mechanical strength
Summary
The DP reflects molecular chain length and is a critical parameter governing solution viscosity, film strength, and processability. High-DP grades are suitable for applications requiring strength, while low-DP grades offer superior ease of handling and coatability.
3. Degree of Hydrolysis of PVOH ―A Parameter Governing Water Resistance, Gas Barrier Performance, and Emulsification
The degree of hydrolysis (DH) represents the percentage of hydroxyl groups relative to the total number of hydroxyl and residual acetyl groups in PVOH, expressed in mol%. It is calculated using the numbers of repeating units “m” (hydroxyl units) and “n” (residual acetyl units) as shown below.
Degree of Hydrolysis (mol%) = 100 × m / (n + m)
Residual acetyl groups are acetyl groups that remain unconverted during hydrolysis. The relationship is:
Residual acetyl groups (mol%) = 100 − Degree of Hydrolysis (mol%)
Accordingly:
- High DH → more hydroxyl groups, fewer residual acetyl groups
- Low DH → fewer hydroxyl groups, more residual acetyl groups
PVOH grades are commonly classified by DH into fully hydrolyzed, intermediately hydrolyzed, and partially hydrolyzed types.
In the production of PVOH, the extent to which acetate groups are replaced with hydroxyl groups during the hydrolysis reaction can be controlled to a certain degree, resulting in a wide range of grades with different DH.
Effects of the Degree of Hydrolysis on Properties
Differences in the DH significantly influence the following properties. Please refer to this information when selecting the appropriate grade for your application.
- Water Solubility: Lower DH result in easier dissolution in water and enable dissolution at lower temperatures. Fully hydrolyzed grades typically require heating to approximately 95 °C for dissolution. (However, if the DH becomes too low, the increased influence of residual hydrophobic acetate groups can reduce water solubility.)
- Gas Barrier Properties: Higher DH provide superior gas barrier performance. Fully hydrolyzed grades are therefore suitable for applications—such as food packaging—where gas barrier properties are required.
- Water Resistance: As the DH increases, the water resistance of dried films improves. Fully hydrolyzed grades are advantageous for applications exposed to moisture.
- Film Formation: The combination of DH and DP determines film strength, flexibility, and transparency.
- Surface Activity and Emulsification Stability: Partially hydrolyzed grades contain hydrophobic acetyl groups as well as hydrophilic hydroxyl groups, resulting in lower surface tension and making them suitable for use as emulsifiers and dispersants
Evaluation points:
water dissolution temperature and rate, oxygen transmission rate (OTR), water resistance (under water contact conditions), emulsion stability
Summary
The DH, expressed as the mol% of hydroxyl groups, plays a crucial role in defining water solubility, water resistance, and gas barrier performance. By optimizing the combination of DH and DP, a wide range of material designs can be achieved to meet diverse application requirements.
Table 1: Relationship between physical properties and degree of polymerization / degree of hydrolysis
How to read the table:
↑ = increases / becomes stronger
↓ = decreases / becomes weaker
Double arrows (↑↑ / ↓↓) indicate a stronger degree of influence.
| DH | DP | |||
Low | High | Low | High | ||
Strongly influenced by the DP | Aqueous solution viscosity | ↓ | ↑ | ↓↓ | ↑↑ |
Film properties | ↓ | ↑ | ↓↓ | ↑↑ | |
Strongly influenced by the DH | Solubility, Dissolution rate | ↑↑ | ↓↓ | ↑ | ↓ |
Water resistance | ↓↓ | ↑↑ | ↓ | ↑ | |
Viscosity stability | ↑↑ | ↓↓ | ↑ | ↓ | |
Thermoplastic moldability | ↑↑ | ↓↓ | ↑ | ↓ | |
Surface activity | ↑↑ | ↓↓ | ↑ | ↓ | |
Dispersibility with other materials | ↑↑ | ↓↓ | ↑ | ↓ | |
Compatibility with starch | ↑↑ | ↓↓ | ↑ | ↓ | |
4. High Water-Soluble PVOH and Modified PVOH
Conventional PVOH generally requires heating to around 95 °C for dissolution. In recent years, however, there has been a growing demand for PVOH that can dissolve in lower‑temperature water, driven by the need to simplify formulation processes and reduce environmental impact through improved energy efficiency. Lower-temperature solubility is also important for coating heat-sensitive substrates and for applications requiring rapid dissolution, such as packaging films and binders.
For such applications, partially hydrolyzed and modified PVOH grades are commonly used.
Kuraray’s High Water-Soluble Grades
Kuraray offers a lineup of high water-soluble PVOH grades tailored to specific application needs:
- KURARAY POVAL™ 5-74
A low-degree-of-polymerization, partially hydrolyzed grade with a certain level of solubility under room‑temperature water conditions, suitable for low-viscosity applications. - KURARAY POVAL™ 25-88 KL
A modified grade that combines high water solubility with moderate film strength, offering broad versatility.
Key Considerations When Using High Water-Soluble Grades
While high water-soluble grades provide improved solubility, their water resistance after drying is generally lower than that of standard grades. When both high-water solubility and water resistance are required, formulations incorporating crosslinking agents can be effective. Kuraray also provides formulation support tailored to specific applications and areas of use.
For assistance with grade selection or formulation design, please feel free to contact Kuraray’s technical team via the inquiry form on this website.
Evaluation points:
High water solubility (dissolution temperature and time), water resistance of the dried film, compatibility with crosslinking agents
Summary
High water soluble PVOH grades are effective for simplifying processing, but understanding the trade-off with water resistance is essential. Combining these grades with appropriate crosslinking systems can enable both solubility and durability.

